|
Choice should be based on the most suitable device for the patient ideally assessed using the In-Check® DIAL G16.Management of Acute Asthma in Primary Care Oct 2018 (Norfolk & Waveney) review dueĬhronic Obstructive Pulmonary Disease (COPD) Primary Care Guideline Aug 22 update (Norfolk & Waveney)ĬOPD Rescue Packs: Quick Reference Guide Sep 2019 (Norfolk & Waveney) -review dueĮnsure patients have a self-management action plan They are based on national guidelines with additional comments and advice from local respiratory specialists.Īsthma Primary Care Guideline Jan 2021 ( Norfolk & Waveney) inhaler review in progress - for inhaler choice see updated Asthma inhaler types and devices link below OR relevant net.formulary subchapter These guidelines are written in conjunction with The Norfolk & Waveney Respiratory Working Group. 2017 389(10082):1919–1929.Formulary Chapter 3: Respiratory system - Full Chapterįor the diagnosis and treatment of asthma & COPD please refer to Norfolk & Waveney Primary Care Guidelines Single inhaler extrafine triple therapy versus long-acting muscarinic antagonist therapy for chronic obstructive pulmonary disease (TRINITY): a double-blind, parallel group, randomised controlled trial. Single inhaler triple therapy versus inhaled corticosteroid plus long-acting β2-agonist therapy for chronic obstructive pulmonary disease (TRILOGY): a double-blind, parallel group, randomised controlled trial. Extrafine inhaled triple therapy versus dual bronchodilator therapy in chronic obstructive pulmonary disease (TRIBUTE): a double-blind, parallel group, randomised controlled trial. Indacaterolglycopyrronium versus salmeterol-fluticasone for COPD. 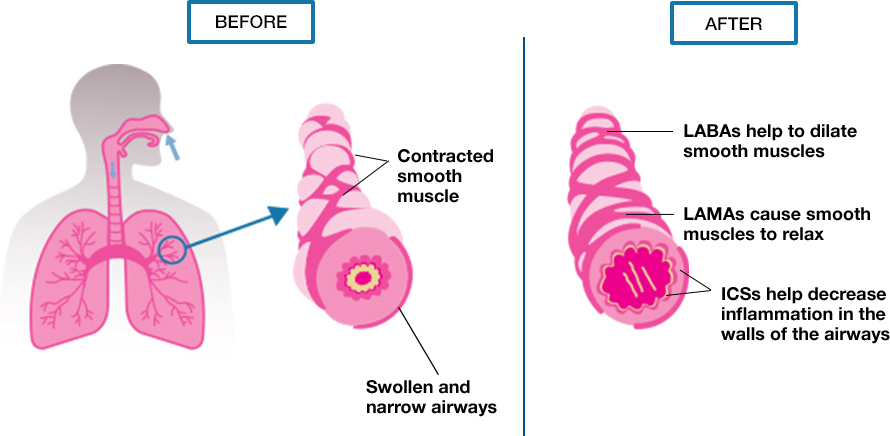
Wedzicha JA, Banerji D, Chapman KR, et al. Global Initiative for Chronic Obstructive Lung Disease Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease. The authors report no other conflicts of interest in this work. BC reports grants to the Division of Pulmonary and Critical Care from AstraZeneca, and that he is a consultant for GlaxoSmithKline, Boehringer-Ingelheim, AstraZeneca, Novartis, Pulmonix, Chiesi, and Menarini, all outside the submitted work. SP is employed by Chiesi, the sponsor of TRILOGY, TRINITY, and TRIBUTE. AP reports grants, personal fees, nonfinancial support, advisory board membership and consultancy work from Chiesi, AstraZeneca, GlaxoSmithKline, Boehringer-Ingelheim, Mundipharma, and TEVA, personal fees and nonfinancial support from Menarini, Novartis, and Zambon, and grants from Sanofi, all outside the submitted work. LMF reports grants, personal fees, and nonfinancial support from Boehringer-Ingelheim, Chiesi Farmaceutici, GlaxoSmithKline, Merck Sharp & Dohme, Takeda, AstraZeneca, Novartis, Menarini, Laboratori Guidotti, and Almirall, personal fees and nonfinancial support from Pearl Therapeutics, Mundipharma, and Boston Scientific, personal fees from Kyorin, Bayer, and Zambon, and grants from Pfizer, Dompè, Malesci, Alfasigma, and Vree Health Italia, all outside the submitted work. Ongoing studies are examining the role of triple therapy in less severe symptomatic patients with COPD and asthma-COPD overlap.Īdrenergic β2 receptor agonists chronic obstructive exacerbations glucocorticoids muscarinic antagonists pulmonary disease review safety.ĭisclosure LV reports grants and personal fees from AstraZeneca, grants from Philips, and Fisher and Paykel, and personal fees from Chiesi, GSK, Pulmonx, Menarini, and Boehringer-Ingelheim, all outside the submitted work. The evidence suggests that triple therapy is the most effective treatment in moderate/severe symptomatic patients with COPD at risk of exacerbations, with marginal if any risk of side effects including pneumonia. Furthermore, triple therapy showed a promising signal in terms of improved survival. Overall, compared to LAMA, ICS/LABA, or LAMA/LABA, triple therapy decreased the risk of exacerbations and improved lung function and health status, with a favorable benefit-to-harm ratio. Of the five main fixed triple studies completed so far, three evaluated the efficacy and safety of an extrafine formulation of beclometasone dipropionate, formoterol fumarate, and glycopyrronium the other two studies evaluated fluticasone furoate, vilanterol, and umeclidinium. This review presents the clinical evidence that led to the approval of these triple therapies, discusses the role of ICS in patients with COPD, and presents data on the relative efficacy of "fixed triple" (ICS/LAMA/LABA) therapy vs LAMA, ICS/LABA, and LAMA/LABA combinations, and summarizes studies in which ICS/LABAs were combined with LAMAs to form "open triple" combinations. 
Recently, two "fixed triple" single-inhaler combinations of an inhaled corticosteroid (ICS), a long-acting β 2-agonist (LABA), and a long-acting muscarinic antagonist (LAMA) have become available for patients with COPD.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
